How to make a lemon battery light a LED bulb
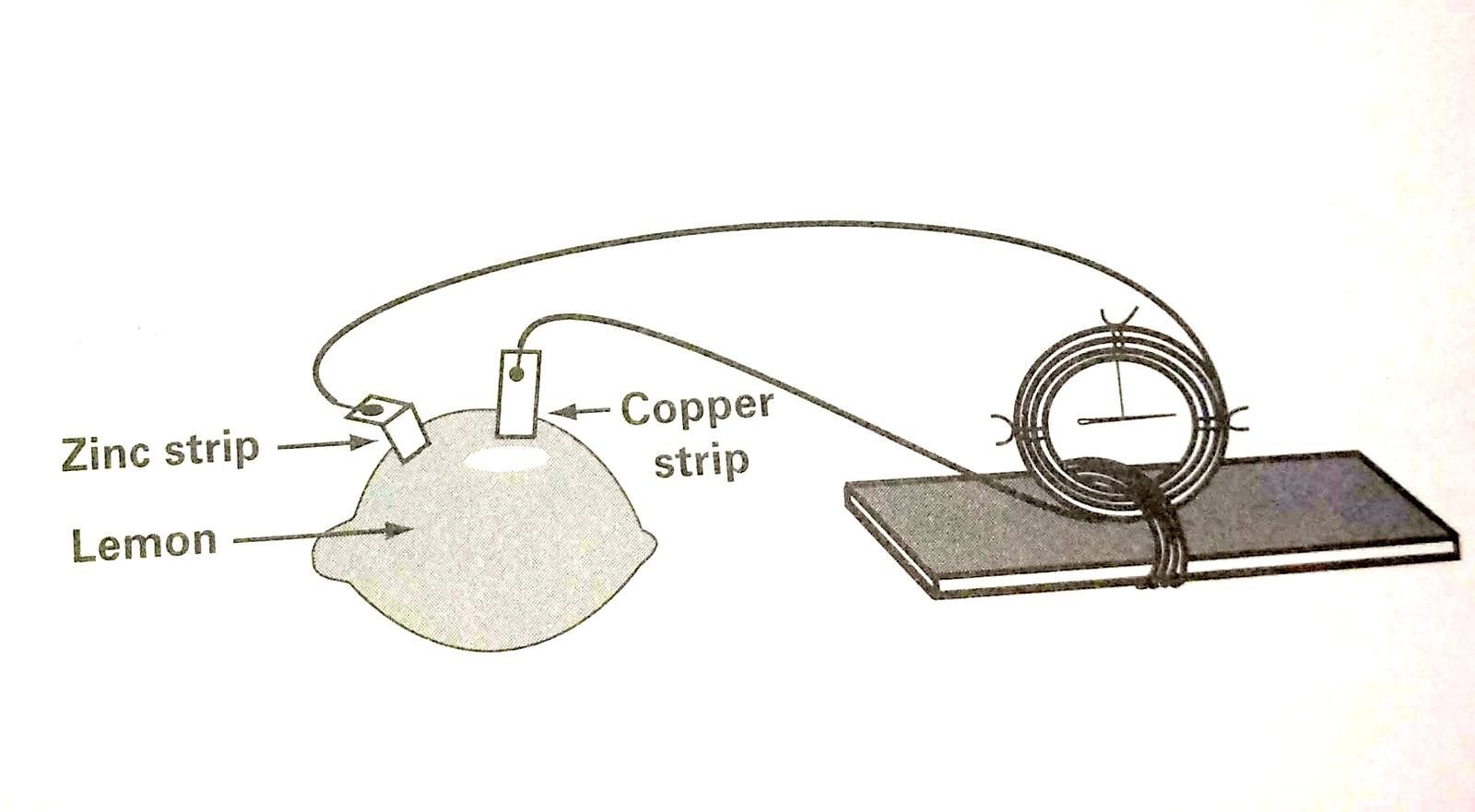
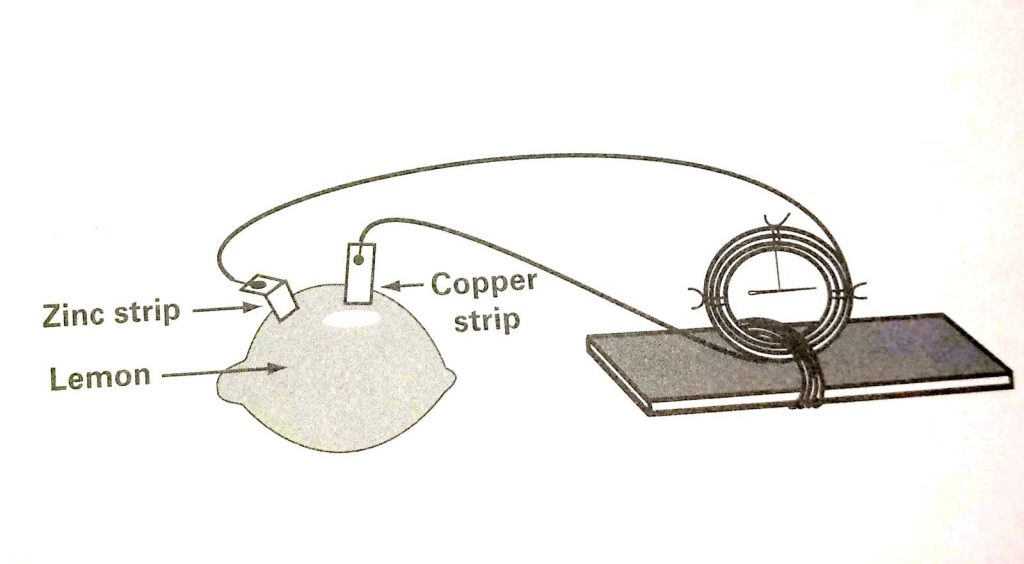
How to make a lemon battery A lemon battery is made with lemon and two metallic such as copper and zinc. Copper and zinc work well together with lemon to make lemon battery because lemon contain citric acid similarly Tomatoes and other citrus fruits contain enough acid to effect the galvanometer.
How to make a lemon battery in an easy way you don’t need too many material.it is very simple to make,At first you have to keep this thing in mind that you can not run any motor or large bulb with this battery because lemon battery can only glow a dim light.
Note;make sure that you are experimenting under the supervision of adults.
Material Needed for How to make lemon battery
- Large fresh and juicy lemon
- Zinc and copper strip
- Galvanometer
- Knife
Procedure how to make lemon battery
- Just for fun, let’s try to learn how to make lemon battery?
- Make two small slits in the lemon, close together, with the knife.
- Attach the copper strip to one wire of the galvanometer and insert it into the slits in the lemon.
- Push the zinc strip into the other slit. The zinc strip must not touch the copper strip. There should be reasonable distance between these two if you want to make lemon battery in perfect way.
- Wait for few seconds and then observe the needle on the galvanometer at the same time you should touch the wire with zinc strip. What happened? Wait a few seconds more and try again.
- Using information you learned in activities 8.17 and 8.18, explain what is happening.
- You can use other citric fruits like tomato and grapes
How to make lemon battery: For critical thinker: flow of electrons can be possible with many chemicals. Not only the citric fruit but you can use the juice of these fruits as well. you can try a penny in the place of the copper strip to make lemon battery and a silver dime (you’ll have to find one that’s several year old to get silver) instead of the zinc.
Take it from there and continue your exploration to make lemon battery. Make sure to write each and every thing you use to make lemon battery and then what happened ? how did you find this activity ?so that you can remember and be able to share your information.
Skills; Observing, inferring, communicating, comparing and contrasting, formulating hypotheses, identifying and controlling variables, experiment