Grade 5 Science Fair Project fireproof balloon

Grade 5 Science Fair Project fireproof balloon
Purpose :
To observe the phenomenon of heat absorption and transfer of energy using water , the air inside a balloon, and an external flame.
Balloons are rather fragile things. When the air inside a balloon is released suddenly, there will be an explosion. The pop sound is caused by the rapid contraction of the latex, which produces ripping friction that breaks the sound barrier.
Therefore, balloons should be kept away from needles, and other sharp objects. They also need to be kept away from flames. A fire can weaken the rubber and cause it to burst.
In this experiment you will find out how you can hold a balloon directly over a flame without breaking the balloon.
It takes ten times as much heat to raise the temperature of 1 gram of water by 1degree Celsius than it does to raise the temperature of 1 gram of iron by the same amount.
This is why it takes so long to bring a teakettle of water to the boil. On the other hand, when water cools, it releases a great deal of heat. This is why areas near oceans or other large bodies of water do not get as cold in winter as areas at the same latitude further inland.
Important Notice : Practice caution when handling matches. Avoid burning yourself and causing fire accidents.
Hypothesis :
The Balloon will not explode when it is filled with water and placed over a flame.
Variables :
- Strength of latex
- Amount of water
- Flame size
- Time
Materials Needed :
- two balloons.
- several matches
- water
Procedure :
- Inflate one of the balloons and tie it.
- Place 60 milliliters (¼ cup) of water in the other balloon, and then inflate it and tie it shut.
- Light a match and hold it under the first balloon. Allow the flame to touch the balloon. What happens? The balloon breaks, perhaps even before the flame touches it.
- Light another match. Hold it directly under the water in the second balloon. Allow the flame to touch the balloon. What happens with this balloon? The balloon doesn’t break. You may even see a black patch of soot form on the outside of the balloon above the flame.
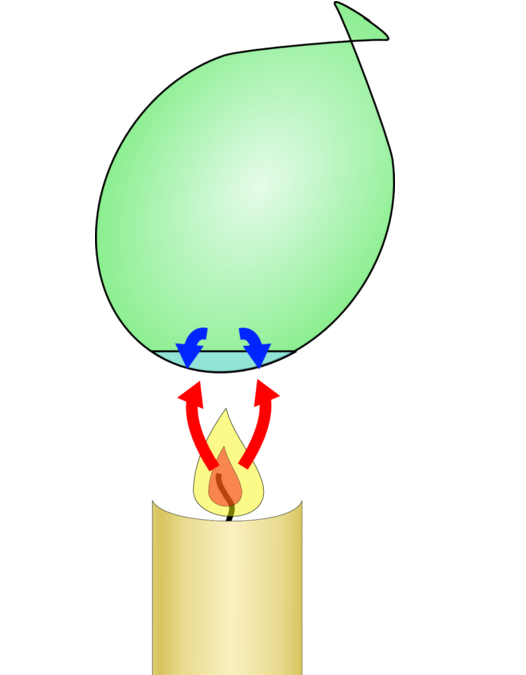
Summary of results
When water inside the balloon is placed in the flame, the water absorbs most of the heat from the flame. Then, the rubber of the balloon does not become very hot. Because the rubber does not become hot, it does not weaken, and the balloon does not break.
Water is a good absorber of heat. It takes a lot of heat to change the temperature of water.
Google Science Journal App an Excellent tool for Science Fair Project Research
Which colors absorb more sunlight than others? Science Fair Projects for kids
Science fair Project grade level 4 to 6 topic Seeds and Needs







